Chronic illness patients often struggle to keep up with medications that need frequent, timely doses. What if a single shot lasted for months?
On average, patients with chronic illnesses follow their prescribed treatments about 50 percent of the time. That’s a problem. If drugs aren’t taken regularly, on time, and in the right doses, the treatment may not work, and the person’s condition can worsen.
The issue isn’t that people are unwilling to take their prescriptions. It’s that some drugs, like HIV medications, require unwavering commitment. And essential medicines, like insulin, can be brutally expensive. Plus, the Covid pandemic illustrated the difficulties of delivering perishable follow-up vaccine shots to regions with no cold chain. “Are we really squeezing all the utility out of those drugs and vaccines?” asks Kevin McHugh, a bioengineer at Rice University. “The answer is, in general, no. And sometimes we’re missing out on a lot.”
For example, the injectable drug bevacizumab can be used to treat macular degeneration, a leading cause of blindness. But even though it’s effective, dosing adherence is notoriously low. “People hate getting injections into their eyes,” McHugh says. “And I don’t blame them at all—that’s terrible.”
McHugh’s lab is in the drug delivery business. The goal is to give patients what they want—less hassle—while also giving them what they need: consistent dosing. The lab’s answer is an injection of drug-delivering microparticles that release their contents in timed delays that can span days or even weeks. “We’re trying to engineer these delivery systems to work in the real world, as opposed to in this idealized version of the world,” McHugh says.
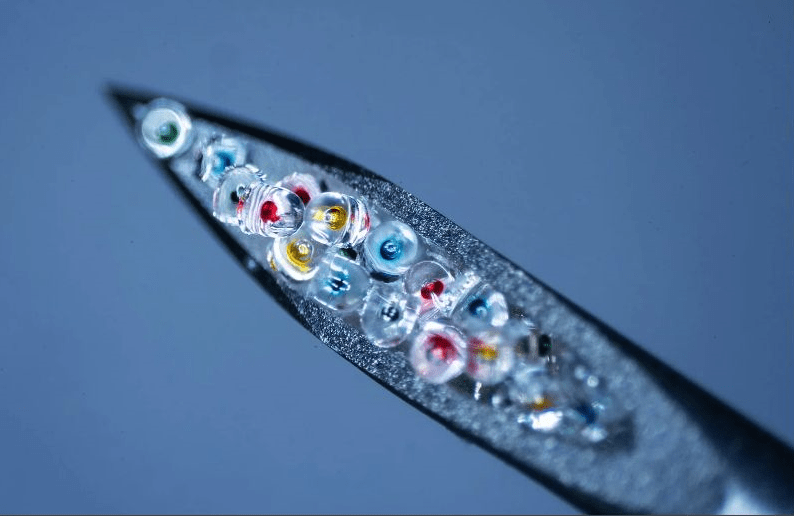
In the June issue of Advanced Materials, McHugh’s team described how their system works. It starts with an injection containing hundreds of tiny microplastic particles, each encapsulating a small dose of a drug. These miniscule capsules are made of the polymer PLGA, which our bodies break down safely. By adjusting the molecular weight of the polymer used for each capsule, the scientists can control how fast they erode and release medication. In this study, the team demonstrated a single shot containing four groups of microparticles that released their contents at 10, 15, 17, and 36 days after injection.
“Having long-acting delivery strategies is a great unmet need,” says SriniVas Sadda, an ophthalmologist with UCLA and the Doheny Eye Institute who was not involved in the study. The patients Sadda sees are elderly. They are often dependent on family members for transportation and may skip appointments because of other health problems. “Maybe they’ve fallen and broke their hip and they end up not coming in,” he says. “Missed visits can be a big problem because you miss treatment and the disease could get worse. And it’s not always possible to recover.”
It’s hard to have delicate control over the levels of a drug in your body, in part because most medications operate like sledgehammers. Pop an ibuprofen or an antidepressant, and those levels will spike as the drug quickly passes through your gastrointestinal tract. Extended release pills prolong a drug’s effect but still taper off from a peak. And you can’t simply front-load a steep dose to delay the next one, since some drugs, like insulin, have a narrow “therapeutic window” between being helpful and dangerous.
Ironically, new and more advanced kinds of drugs have only made this problem more daunting. In 2021, seven of the 10 best-selling drugs in the United States were biologics, a class that includes proteins, hormones, and gene therapies. Biologics are more finicky than small molecules like ibuprofen, and rarely work orally. But they’re effective. “The potency and the specificity provided by protein drugs like antibodies is so great,” McHugh says. “Now the question would be how to make them last for long.”
During a postdoctoral fellowship at MIT about six years ago, McHugh experimented with manipulating polymers to encase drugs. His team invented a type of microparticle that encapsulated a drug using PLGA because the polymer has been used clinically in FDA-approved treatments since 1989. It was clear that changing the polymer’s molecular weight would delay its degradation—and the drug’s release—but the technique was expensive and hard to scale up. And some of the most important applications, like for vaccines, need to be extremely low-cost. “If we’re trying to develop and deliver vaccines in low- and middle-income countries, maybe these technologies have to cost a couple of pennies,” he says. “How do we make a billion of these?”
So when McHugh started his own lab at Rice, his team put his original process under the microscope. His previous method entailed casting a microscopic PLGA “bucket” to fill with a drug, then adding a flat “lid” of the polymer. They’d line the bucket and lid up under a specialized microscope, smush them together, and heat them to form a seal. Too many steps, McHugh thought.
He asked Tyler Graf, the PhD candidate leading the project, whether they could instead dip the unsealed particles—en masse—into a melted pool of PLGA. Intrigued, Graf tried. No dice. The individual buckets couldn’t form clean seals because the PLGA wouldn’t break off from the pool. Long strings of the polymer dragged out, like cheese pulling off of pizza. “That’s obviously not feasible because that’s extra material that can’t fit through a needle,” says McHugh.
Graf wondered what would happen if they nixed that step entirely. He took a glass slide dotted with barely visible unsealed buckets and flipped it face down above a hot plate. The top of each bucket pinched in and sealed off. “We got a little bit lucky,” McHugh says. “That was the first place where we were thinking this is really going to be something exciting here.”
Today they use lab robots to fill the capsules, and they are working to automate the entire process, which they call Pulsed, for Particles Uniformly Liquified and Sealed to Encapsulate Drugs. McHugh believes that this automation cuts costs and makes the tech scalable. Thanks to minor tweaks to the capsule recipe, the Pulsed particles rupture with distinct, predictable delays, ranging from days to over a month.
For their recent study, their team wanted to know how quickly these capsules would degrade in a living animal, so they compared the timing in test tubes to that in mice. In one trial, they loaded the microparticles with tiny fluorescent molecules in lieu of medicine. With the mice, they injected a small volume of the capsules beneath the animals’ skin, then they tracked the fluorescence as the molecules diffused outward. With the test tubes, they kept the capsules in a saline solution at body temperature and checked to see when the fluorescent molecules spilled into the solution. In all cases, the timing matched. This means that timing predictions based on lab experiments will likely hold up well in living bodies.
Continue reading the full article at Wired
Enter here to access the original report: